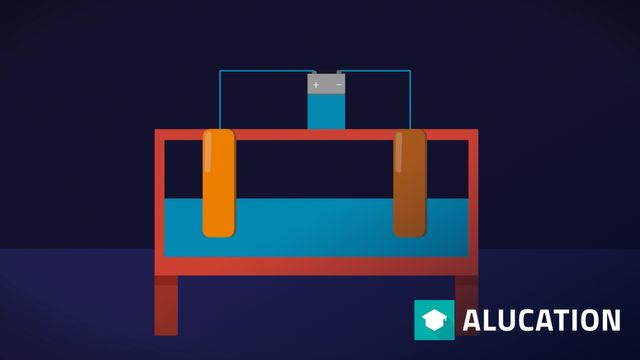
Q 12 Concentration-dependent redox potentials
The concentration dependence of the redox potential is explained and Nernst's equation is derived from it. Original: "Wolfgang Dukorn" Original: German. All translations in this video have been done by the alugha AI.