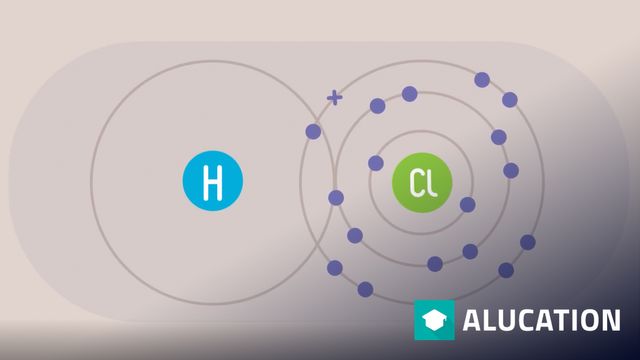
Drawing Electron Configuration Diagrams | Properties of Matter | Chemistry | FuseSchool
Click here to find more videos: https://alugha.com/mysimpleshow Learn the basics about Drawing electron configuration diagrams. Find out more in this video! This Open Educational Resource is free of charge, under a Creative Commons License: Attribution-NonCommercial CC BY-NC ( View License Deed: h