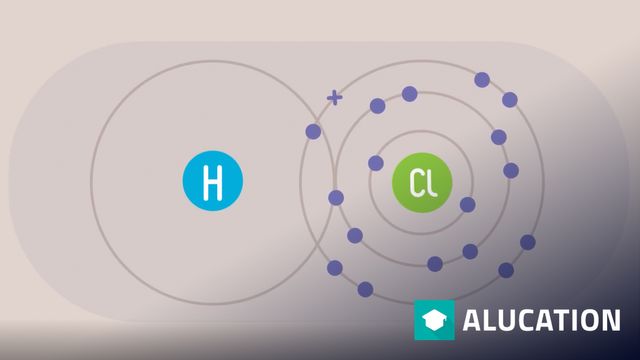
08 The Noble Gas Configuration
The noble gas configuration is a particularly stable electron configuration. The driving force for many chemical phenomena is this state. It should be remembered again that the nucleus is not altered during chemical reactions. Original: "Wolfgang Dukorn" Original: German. All translations in this